








About us

Hengyong Electronic Products Co., Ltd., also known as Dongguan Hengyong Electronic Products Co., Ltd. was established in 1993 as a Taiwan-funded enterprise, registered as the "Shangying" brand. The self-purchased land was built 10,000 m2, and the remote control industry started. In 2009, Hengyong Group successively set up the projector business unit and the medical device business unit. The Group focuses on upgrading medical equipment divisions directly under the medical equipment processing, research and development, design, production, manufacturing and sales. Hire qualified R&D personnel to design and develop import: image imaging research and development, sensor + embedded + algorithm + short-range wireless transmission principle research, ultrasonic conversion thermal effect & cavitation effect & mechanical effect realization principle research and development, optical research and development. Including IC software programming, hardware design, design, structural design, mold design, PCB circuit design, packaging design. For the best cooperation, the efficient delivery process meets the needs of customers. The company invests in related parts factories, making the self-made rate more than 85%. The company has R & D design, mold making, plastic injection molding, fuel injection, printing, hot stamping, ultrasonic, laser engraving, rubber vacuum forming, COB IC, automatic SMD, assembly of finished products and the establishment of a reliable laboratory one-stop service . The company has passed the ISO9001 quality system to effectively control the product quality process. At the same time, in order to comply with WEEE/ROHS/REACH related regulations, the company has passed ISO14001 to establish an environmental testing laboratory, and purchased ICP-0ES heavy metal tester and X-Ray equipment to ensure Products meet ROSH and other related requirements. The company has passed ISO13485 professional medical device products to ensure process management standards on the basis of medical laws and regulations.
Self-made ability to control quality, using imported high-quality raw materials to ensure product life. The company currently has 9 products, and develops 1-2 new products every year. The self-developed medical products have their own patent protection. The self-developed medical products meet all the certification conditions and obtain medical CFDA certification.
The company strictly abides by the quality policy: implementation of preventive correction, corporate self-improvement
The company strictly abides by the quality objectives: improving with customer requirements and aiming at customer satisfaction
The company strictly abides by the business philosophy: focus, innovation, and change of mind
1987---Tongyong Plastic Industry Co., Ltd. was established in Taipei
1993---Invested in Dongguan, Guangdong Province, Hengyong Electronic Products Co., Ltd.
2002---ISO9001-2000 quality assurance system certification
2003---Investment subsidiary Dongguan Jingming Electronic Products Co., Ltd.
2004---Investment in Jiangsu Kunshan Guanzhi Technology Electronics Co., Ltd.
2005---Improve environmental pollution prevention system and establish environmental testing laboratory (ROHS)
2006---ISO14001-2004 Environmental Management System Certification
2007---Imported RF/Bluetooth/WiFi remote control and receiver and related products for development, production and production
2009---Founded directly under the Group Medical Devices Division
2010---Import projector and teaching machine division
2013---Introduction of 100,000 clean room
2015---Introduction of automatic SMT workshop
2018--- passed the ISO13485 professional medical quality legal and regulatory system
| Business type registered capital | Investment capital |
| Sole proprietorship | HKD1625万 / RMB9000万 |
| Plant area | Management system certification |
| 10,000square meter | ISO13485;ISO9001; ISO14001; |
| Mumber of employees | Number of R&D department |
| 350~500 people | R&D team number 60 people |
| QC people | major markets |
| 69 people | China,Japan,Europe |
| Main products | Main customers |
| Medical equipment,big health smart medical equipment. | Lianying,Japan,EU etc. |
| Mechanical monthly capacity | Assembly line |
| 300,000U/M | 12 workshp in dust-free workshop |

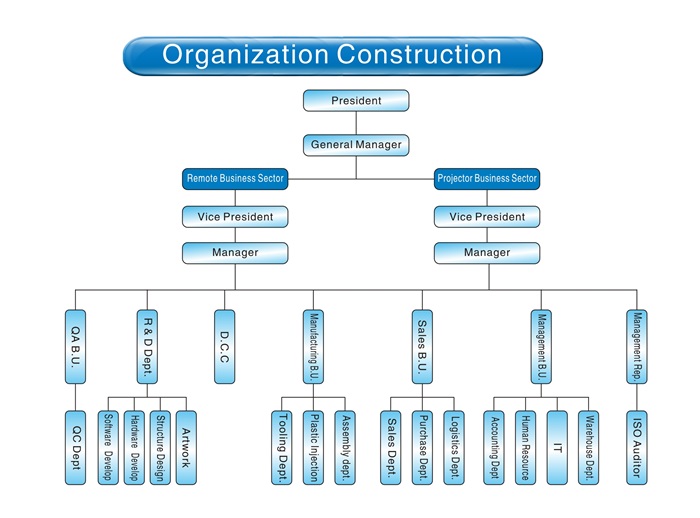
- Undertake ODM, OEM medical equipment, large health intelligent medical
- equipment design and manufacturing, Undertake agents in various provinces
- Contact person:Elissa Jiang
- Mobile:0086-13360689788

